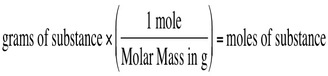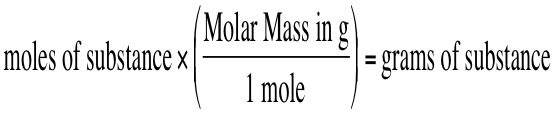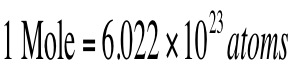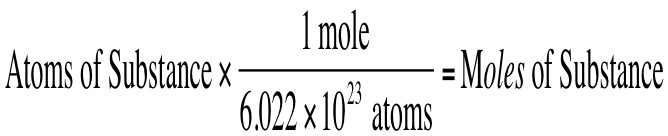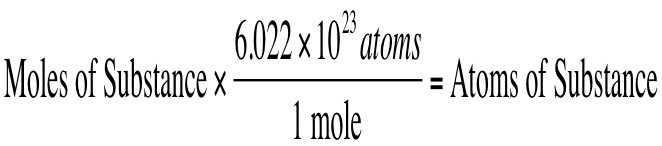District 4 - Stoichiometry and Solutions

This is one of two powerhouse Districts. There is rarely a Hunger Games that does not have these tributes in the final group of individuals fighting to the death. The mere utterance of there name is enough to strike fear into the hearts of their opponents. If there is going to be a District that is going to take you down, this may be the one that does it. It is for this reason that intense training will be necessary if you are to ensure that you are victorious! They have many weaknesses, but these weaknesses can only be exploited if you have adequate training.
Your Training
Training Resources
Chemical Equations
|
The law of conservation of mass explains that matter cannot be created or destroyed, it can only change forms. Therefore, we cannot destroy something. All we can do is change the form that it exists as.
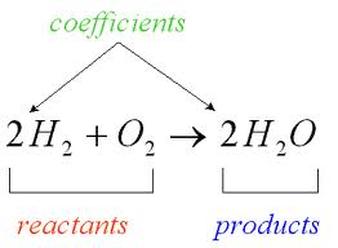
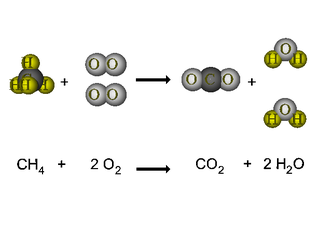
Elements and compounds react to form new elements and compounds. Chemical equations are used to explain a reaction that occurs and what we are left with after a reaction. In a chemical reaction, we have reactants and products. Reactants are to the left of the arrow in a chemical reaction. Products are elements/compounds on the right side of the arrow in a chemical reaction. In a chemical reaction it looks like: Reactants --> Products We can describe the state that a elements/compounds exist in during a chemical reactions. A molecule can be in a solid, liquid, gas, or aqueous state. Aqueous means that the element/compound is dissolved in water. We abbreviate this as follows: (s) = solid, (l) = liquid, (g) = gas, (aq) = aqueous. |
Balancing Chemical Equations
|
Most chemical reactions, as written, do not satisfy the law of conservation of mass. There are more atoms on one side then on the other. Since the law of conservation of mass states that we cannot create/destroy matter, we need to have the same amount on each side of an equation. Therefore, we need the same amount of atoms on each side of an equation to satisfy this law.
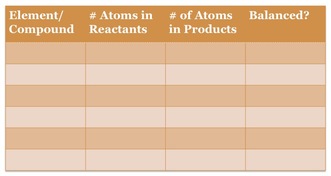
We balance chemical equations so that the number of atoms on the reactants side equals the number of atoms on the products side. We CANNOT change the subscripts. The only thing we can change is the number in front of the element/compound. This is called a coefficient. The best way to balance an equation is the work by filling in the table below. 1. Enter the elements (or compounds if they are present on both sides of the arrow) into the far left column.
2. Count the number of atoms on the products and reactants side and enter into the two middle columns. 3. Finally decide what coefficient you need to place on the reactants and products side in order to make the two middle columns to be equal to one another and enter that into the initial equation. Balancing chemical equations can be very difficult. To get some help visualizing these reactions please go to the pHet simulator linked here and practice balancing equations using this online simulator. |
The Mole
|
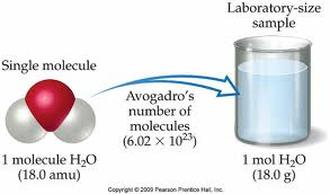
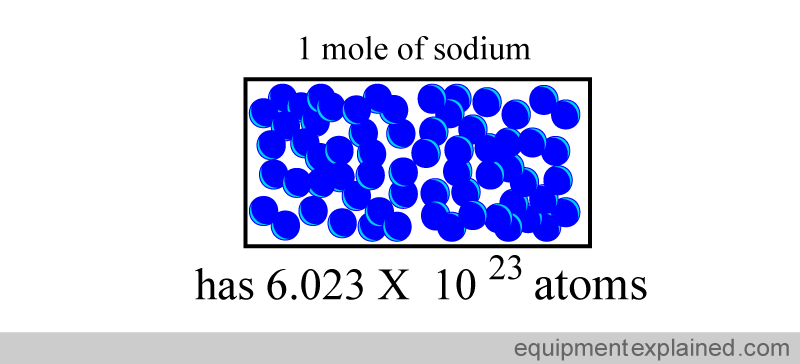
Contained within any substance that we can see is trillions upon trillions of atoms We need a simpler way of conveying the amount of atoms that are contained within any substance The mole is a unit that describes the amount of particles that a substance has. Contained within one mole is 6.022 x 10^23 particles or 602,200,000,000,000,000,000,000 particles!
The mole is simply a number. This is just like the concept of a dozen You can have dozen dogs or dozen chairs, each describes that we have 12 off them. A mole is simply 6.022 x 10^23 of something. This means we could have a mole of atoms or a mole of M & Ms. Each means that we have 6.022 x 10^23 of them. |
Solute and Solvent
|
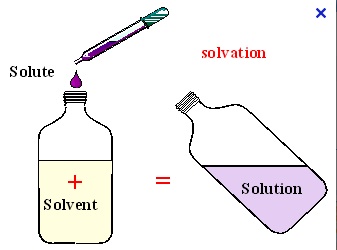
When studying solutions and how to make them, there are two primary ingredients that exist in every chemical solution: the solute and the solvent. The solvent is the substance in which the solute is dissolved. This is the primary ingredient in a solution and it is present in the largest amount. The solute is the substance that is dissolved in the solvent.
It is important to remember that we call water the universal solvent. If ever there is a solution that is water and another compound, then the solvent is always water. When water is the solvent, it is an aqueous solution (which we talked about above). |
Molar Mass, Grams to Moles, and Molarity
|
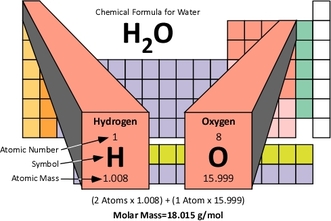
Molar mass or molecular weight is the mass, in grams, of 1 mole of substance. The molar mass is numerically equivalent to the mass that is listed on the Periodic Table. Molar mass is expressed in units of g/mol. So if you wanted to determine the molar mass of carbon, you would find carbon on the Periodic Table and see that its atomic mass is 12, so its atomic mass is 12 g/mol.
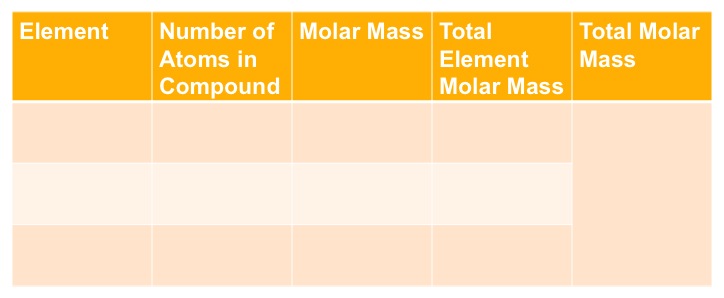
To calculate the molar mass of a compound, begin by drawing the following table and : Once you complete this table you, multiply the number of atoms by the molar mass. This value is the value that goes in the total element molar mass column. Finally, you add all the numbers in the "Total Element Molar Mass" column in order to calculate the total molar mass.
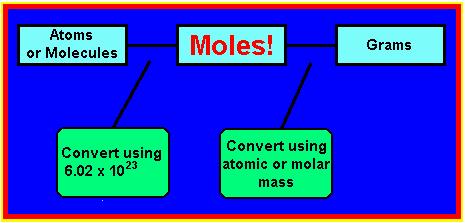
Once you know how to calculate the molar mass you will be able to convert grams of that substance to moles of that of that same substance. We can convert between the two by using the molar mass of the compound. To convert from grams to moles, use the following equation: You also can convert in the other direction by using the reverse equation:
Quick Check! Do These Problems!Knowing how to convert between grams and moles is essential because this allows us to calculate the concentration of a solution. This is important to know once you get into the Arena because the concentration of a solution can be the difference between a solution that you can drink to save your life and a solution that will kill you if you drink it. The concentration of a solution is most often expressed in molarity. Molarity expresses the concentration of solution in terms of moles of solute per litter of solution. Molarity is calculated by: Quick Check! Try These Problems! |
|
Particles to Moles and Molarity
|
|
We also can convert between the number of atoms or molecules of a substance to moles of a substance. Recall, that if we have 1 mole of a substance, then this is the same as saying 6.022 x 10^23 particles. It is important to remember that:
If you know the number of atoms of a given substance, then you can use the following equation to convert from atoms of a substance to moles of a substance:
You also can convert in the other direction (moles to atoms) using the following equation:
Quick Check! Try These Problems! |
Extra Resources
Still confused? Check out these awesome sites: