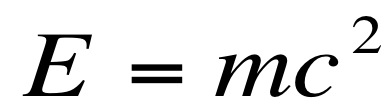District 10 - Nuclear Chemistry

Welcome to your training for how to take down District 10 when you are in the arena. It is no secret that this District possess a power that is unparalleled to the weapons that the other Districts have. In fact, they have the power to vaporize miles of land by splitting only a few kilograms of atoms. District 10 is not to be messed with and they are well aware of this fact. They are advised by arguably the greatest past Hunger Games Champion: Albert Einstein. They possess the ability to create an atomic bomb...so you better be very careful if you choose to do battle with them in the arena.
Past District 10 Champion
Your Training
Useful Files for Your Training
The Fundamental Forces
|
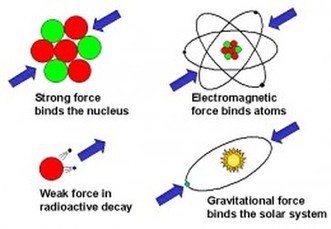
There are 4 fundamental forces in the universe:
An electromagnetic force is an attraction or repulsion between positive and negative charges. This principle is easiest to remember by the phrase, "Like repel and opposites attract." We have a problem though. The nucleus of an atom contains many positive changes (protons). If the nucleus contains a bunch of positive charges, why doesn’t the nucleus of the atom split apart? The strong nuclear force is the force that exists between particles in the nucleus of an atom. This is much stronger then electromagnetic forces, so the attraction between particles in the nucleus is much stronger then the repulsion between protons. The relative strength of the funamental forces is: Gravity < Weak NF < Electromagnetic < Strong NF |
Fission and Fusion
|
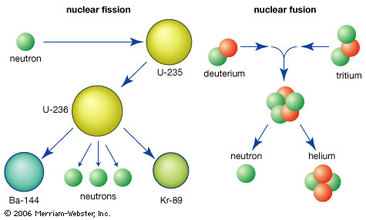
There are two types of nuclear processes that occur at the atomic level: fission and fusion. Fusion describes when two things fuse into one. Nuclear fusion is when two atomic nuclei fuse into one new atom. Nuclear fusion processes are an essentail source of energy in the universe. One of the most common nuclear fusion processes occurs on the Sun. Nuclear fusion is how energy is produced in the Sun.
Fission describes the splitting of something into two Nuclear fission is when a very heavy nucleus splits into a smaller, more stable nucleus. Both fission and fusion release large amounts of energy. This energy that is released is exponentially larger than the energy that is released during a chemical reaction. The energy that is released or absorbed was quantifyed by Albert Einstein in the early years of the 20th century. |
Einstein's Equation
|
The conservation of matter tells us that matter cannot be created or destroyed, it only can change forms. Sometimes, when compounds react the mass of the atom is not the same as the mass of protons and neutrons alone. Since matter cannot be created or destroyed, this mass had to be converted into something else. This matter was turned into energy. Matter can be turned into energy and energy can be turned into matter. Einstein’s equation allows us to calculate the amount of energy that would be released if mass is lost.
In the above equation, E = energy (measured in Joules), m = mass (measured in kilograms), and c = the speed of light( 3.0 x 10^8 m/s or 300,000,000 m/s).
|
Radioactive Decay
|
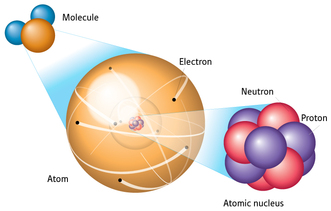
In nature, the same element can have different atomic masses. These are known as isotopes. Isotopes are atoms of the same element have different numbers of neutrons and thus have different atomic masses.
Some atoms are unstable and breakdown. These elements are called radioactive compounds. This process of breaking down an element is known as radioactive decay, which is the spontaneous breakdown of the nucleus into a smaller nucleus, accompanied by the release of particles, radiation, or both. Radioactive decay occurs because the electromagnetic force overcomes the strong nuclear force. When this happens, the positive-positive repulsion of protons is strong then the force that exists between particles in the nucleus, so the atom splits apart and releases particles and/or energy. There are three types of radioactive decay that normally occur:
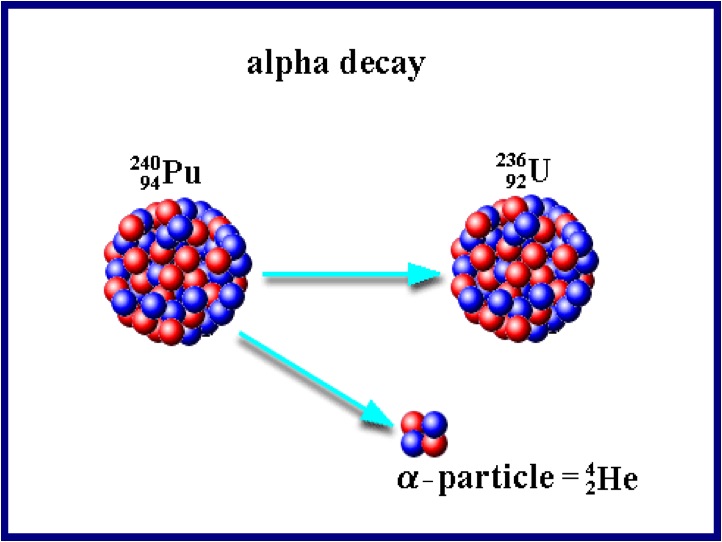
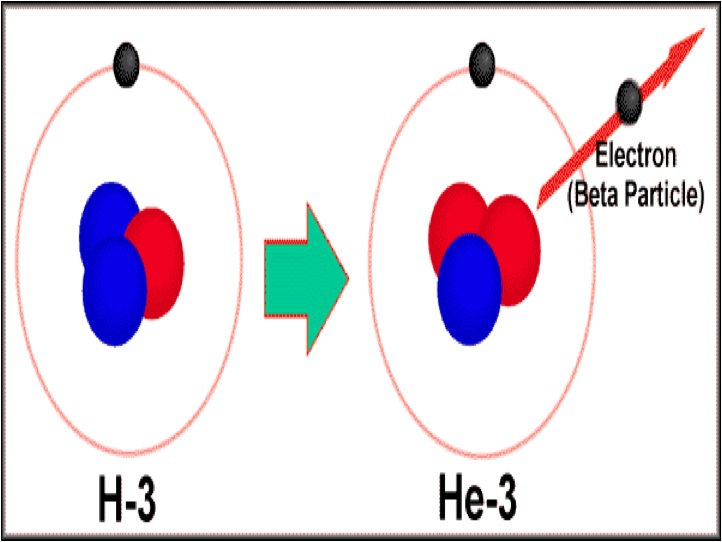
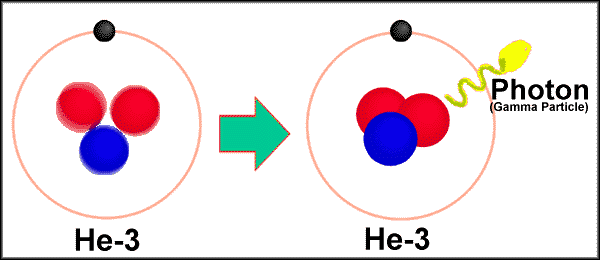
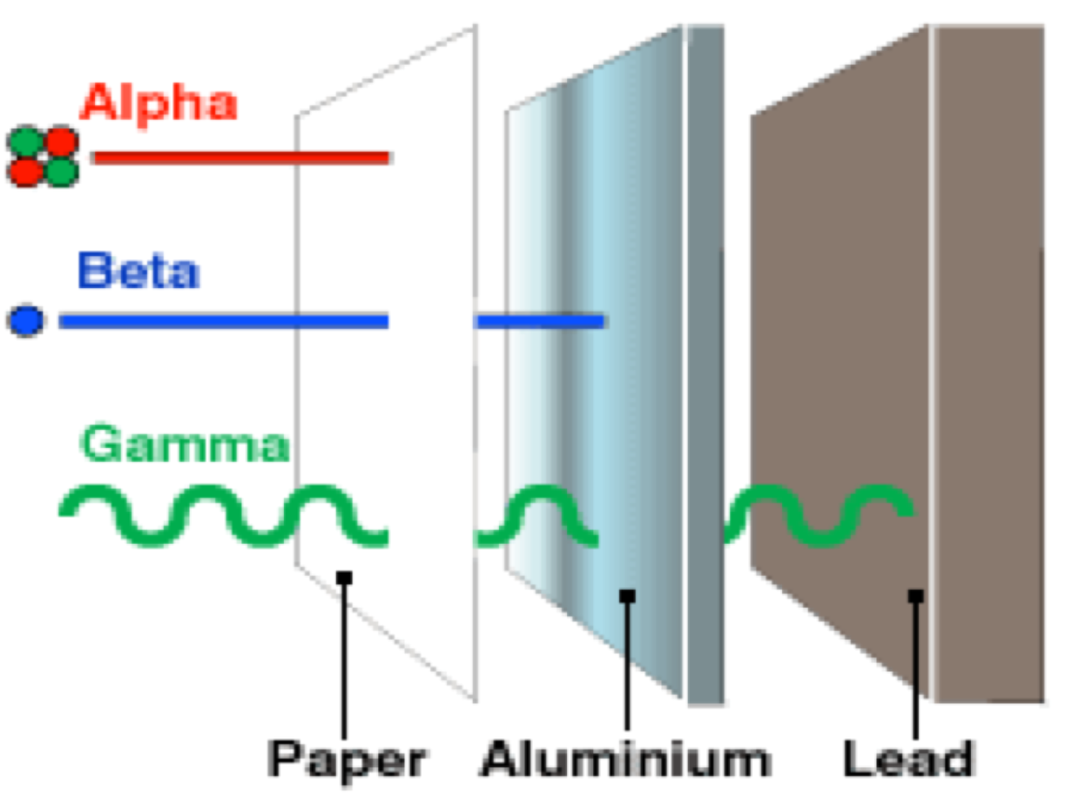
The first type of decay is alpha decay. Alpha decay is when an atom splits its nucleus into a smaller nucleus and a Helium atom (2 protons, 2 neutrons). After alpha decay, we are left with a new atom and one He atom. Alpha particles can only travel a few centimeters in the air. Alpha particles cannot penetrate skin and paper blocks alpha particles. Many elements are unstable because their proton to neutron ratio is too large. Beta decay converts neutrons to protons and releases an electron known as a beta particle. The general equation for beta decay looks like: neutron --> proton + electron Beta particles penetrate much deeper than alpha particles and travel a few meters in the air Most metals provide a shield against beta radiation. Gamma rays are high energy waves that are released from a nucleus when it goes from a high energy state to a low energy state. This type of decay usually occurs along with other types of decay. Gamma radiation is the most dangerous. It can lead to DNA mutations and cancers. Gamma radiation requires thick pieces of lead/concrete in order to stop. |
|
Practice
|
In order to be prepared to do battle with this District, complete the following practice problems:
Answer Key |
A Present from a Sponsor...
|
A sponsor has sent a gift of radioactive isotopes. In order to properly use them to kill District 10, complete the following assessment:
|
Extra Resources
Still confused? Check out these awesome resources!:
- Khan Academy Nuclear Chemistry
- Brightstorm Nuclear Chemistry
- Brainpop Videos (ask Mr. Lance for login information)
- Alpha Decay Simulator
- Beta Decay Simulator