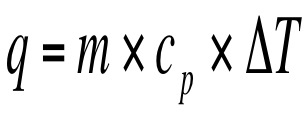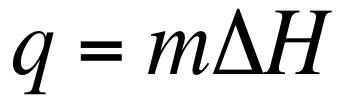District 8 - Thermochemistry

Welcome to your training for how to take down District 8 when you are in the arena. This District possesses the power of fire, so there is no telling how they could harness these skills once they are let loose inside the Arena. This District is in it to win because they are one of only two Districts that has never won the Games. Knowing the skills of this District will be imperative if you hope to kill this District inside the arena. District 8 tributes are capable of killing you and telling you how many calories a kill took once the kill has been completed.
Your Training
Training Resources
Exothermic and Endothermic
|
Heat is the transfer of energy between substances due to a difference in temperature. The faster a substance is moving the higher its temperature will be. A substance at a high temperature has fast moving molecules and a substance at a low temperature has slow moving molecules.
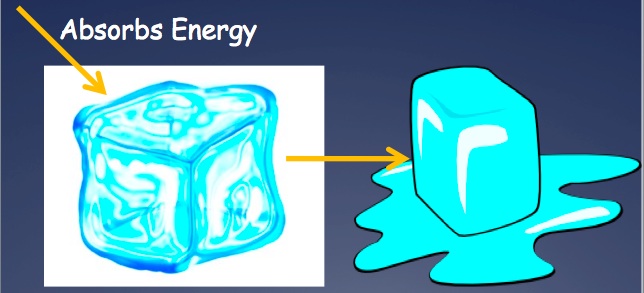
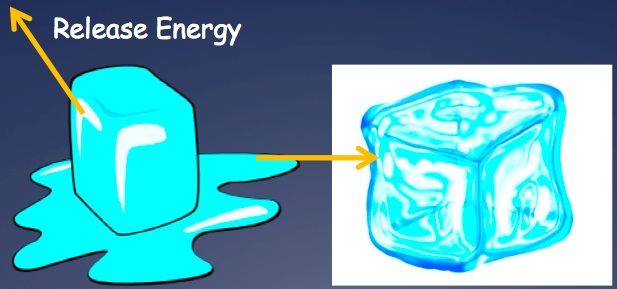
A reaction that releases energy during the reaction is known as an exothermic reaction. Energy is often given off in the form of heat. Energy is listed as a product in the equation: Reactants --> Products + Energy A reaction that requires energy during the reaction is known as an endothermic reaction. Energy often is absorbed in the form of heat, so the substance feels cold. Energy is listed as a reactant in the equation: Energy + Reactants --> Products Phase changes can be classified as endothermic or exothermic. Since you have to add energy to go from a solid to a liquid and a liquid to a gas, these are listed as endothermic processes. Since you have to release energy to go from a gas to a liquid and a liquid to a gas, these are listed as exothermic processes. |
Entropy and Enthalpy
|
There are two main values that are of central concern for thermochemistry. They are enthalpy (H) and entropy (S). Enthalpy is a measure of the total energy that a system has. This value describe the amount of energy released or absorbed during a process/reaction. It is measured in kilojoules (kJ).
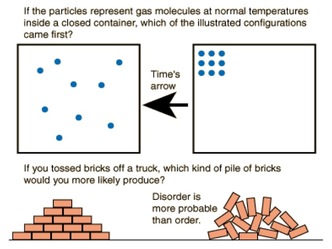
Entropy is based on the fact that the universe tends towards disorder. Under all circumstances, disorder is more favorable than order. The measurement of the randomness or disorder of a system in known as entropy. As a system gains more energy, molecules move faster, and the overall disorder increases. We measure entropy in kJ/mole. |
Phase Diagrams
|
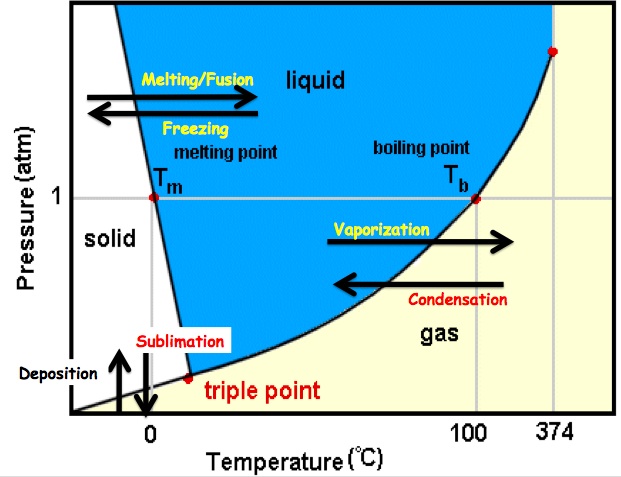
The state of matter that a substance exists in depends on both the temperature and the pressure A phase diagram is a graph of the relationship between the state of the substance and its temperature and pressure. Each substance has its own unique phase diagram.
There are two important points on a phase diagram that can be identified. The triple Point is the temperature and pressure conditions at which the solid, liquid, and gas phases of a substance coexist at equilibrium The critical Point is the temperature and pressure at which the gas and liquid state become identical and form one phase. There are three different regions on a phase diagram: solid, liquid, and gas. The lines that seperate each of these regions from the other regions represent phase transitions. When you cross a line (either by changing temperature and/or pressure), the substance undergoes a phase transition. Some important phase transition vocabulary is listed below:
|
Heating Curves
|
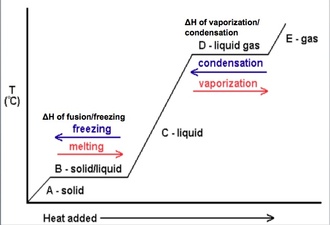
There is a large amount of energy that is required to change a solid into a liquid and a liquid into a gas (and vice versa). The energy that is required to change phases is known as the ΔH of phase change.
We can graph the relationship between temperature and time. This graph between temeprature and time (or heat added depending on the graph) is known as a heating curve. This allows us to identify the areas of melting/freezing and boiling/condensation. Areas of positive slope represent the solid, liquid or gas phases. Areas of no slope represent a phase transition. |
Calorimetry
|
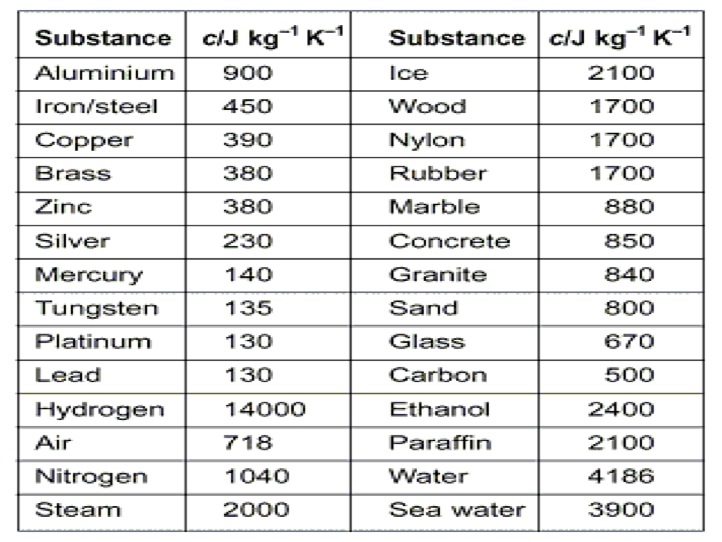
The amount of energy that is transferred to a material is dependent on the nature of the material receiving the energy. We can quantify the amount of heat transferred using the specific heat. The specific heat is the amount of energy required to raise the temperature of one gram of substance one degree Celsius. The specific heat can be represented by the symbol Cp.
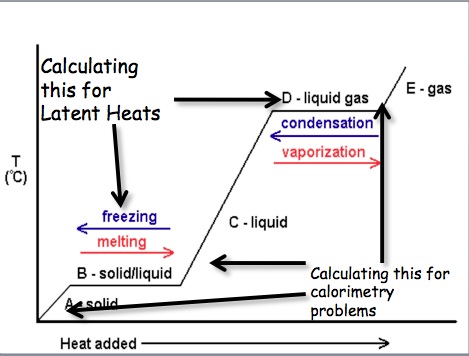
We can measure the amount of heat that is released or absorbed during a chemical process. The process of measuring this change is known as calorimetry. Calorimetry is the science of measuring the heat of a chemical reaction. In the equation q represents the heat gained/released, Cp is the specific heat, and ΔT = Final Temperature – Initial Temp. On a heating curve, these are the areas of positive slope.
We can also calculate the amount of energy that is necessary to convert a substance from one phase to another. This is known as calculating the latent heat. The latent heat is the energy absorbed as a substance changes phases. In the above equation m = mass of compound and ΔH = Enthalpy values for phase changes (constants). On a heating curve these are the values for the areas of no slope.
|
Practice
|
Below you will find problems that will ensure a fast kill of the District 8 tributes:
Answer Key: |
Fire!
Extra Resources
Still confused? Check out these awesome resources!: