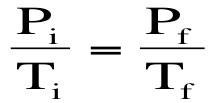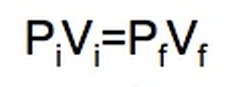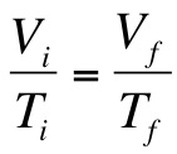District 6 - Gases

Welcome to your training for how to take down District 6 when you are in the arena. The gases are one of the major players in these Games. The gas laws are central to so many things that occur throughout the universe and this District has a very intimate knowledge of how gases can be manipulated for their own advantage. With the advisers Boyle, Gay-Lussac, and Charles, you can expect that this District will go far during the Games. The ability to crush a human being without ever touching them is one of their signature kill moves.
Past District 6 Champions
Your Training
Training Resources
Pressure
|
Gases are one of three states of matter. They move in constant random motion. They fill an entire container and are spread very far apart from one another.
The collisions of gas molecules with a surface are what creates pressure. Pressure the amount of force that is exerted per unit area of surface. Pressure is measured with a barometer and it is measured in many different types of units. The units of pressure are atm, bar, mm Hg, Pa, and torr. |
Diffusion
|
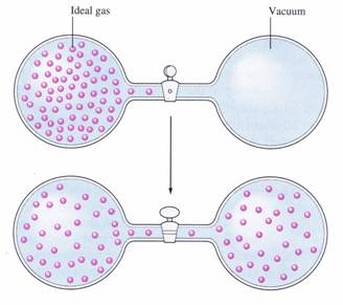
Diffusion is the movement of particles from regions of higher density to regions of lower density. Because gases are constantly moving in random motion, they spread out from one another very quickly. The random motion of gases is what explains the process of diffusion and the rate at which gases diffuse. Gases move very fast. Therefore, gases diffuse through a room at a relatively fast rate.
|
Kelvin and Absolute Zero
|
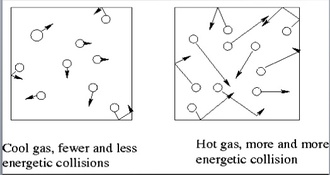
The temperature that we feel is created by the movement of atoms. The movement of gas molecules in the air creates the observable temperature that we experience each day.
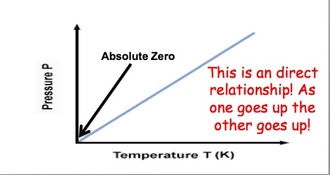
The 2 main temperature scales in chemistry are the Celsius and Kelvin scales The Celsius scale sets the freezing point of water at 0 C and the boiling point of water at 100 C. All other temperatures are based on these two values. The other unit of temperature used in chemistry is the Kelvin scale. It is the official SI unit for temperature. The Kelvin scale sets 0 K as being absolute zero. Absolute zero is the temperature at which all molecular motion stops. This is the coldest possible temperature Temperature is based upon the movement of molecules. Thus, it is impossible to go below absolute zero (0 K) because molecular motion has stopped at this point. To convert from Celsius to Kelvin, we use the equation: Kelvin = Celsius + 273 To convert from Kelvin to Celsius, we use the equation: Celsius = Kelvin – 273 |
Gas Laws
|
We can describe how a gas will change based on the volume, pressure, temperature, and moles of the gas. The gas laws focus on these properties and how changing one affects the other three.
Gay Lussac's Law: Gas molecules move quicker the more we heat them up and increase the temperature. This means that they will be hitting the sides of the container more often and the pressure will also increase as the temperature increases. If we keep volume constant, then pressure and temperature are directly related. Gay-Lussac’s Law relates pressure and temperature through a math expression: Where P represents pressure and T represents temperature. i and f simply mean before and after the reaction.
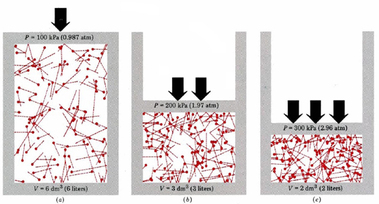
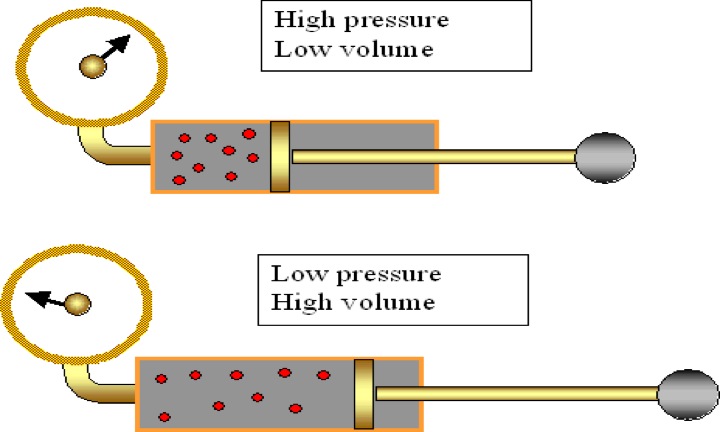
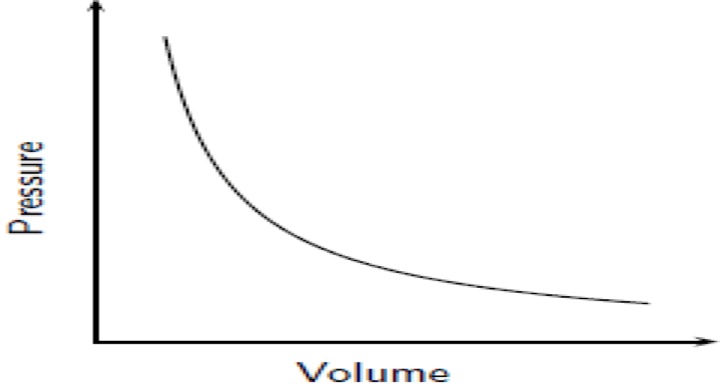
Boyle's Law: If we keep temperature constant, then volume and pressure are inversely related. As one goes up the other goes down If the container gets larger the gas molecules are hitting the walls at a less frequent rate. Therefore, by increasing the volume of a container, we are decreasing the pressure that is felt by that container. The reverse is true if we decrease the volume of a container. Boyle’s Law states that pressure and volume are inversely proportional and they can be described by the equation: Charles Law:
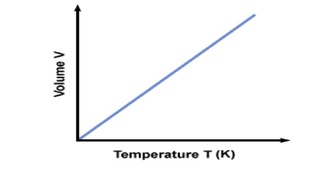
Boyle’s Law looked at the relationship between volume and pressure. Gay-Lussac’s Law looked at the relationship between pressure and temperature Charles Law focuses on the relationship between temperature and volume. As you heat up gas molecules they move quicker and this causes them to hit the sides of the container with greater frequency. If the container is capable of expanding, so that pressure can remain constant, then the volume will increase as the temperature increases. Charles Law can be described by the equation: |
|
Ideal Gas Law
|
STP:
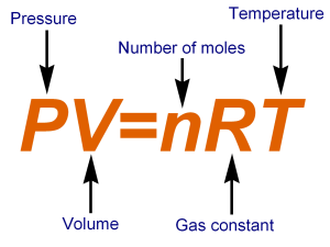
Scientists use the idea of standard temperature and pressure (STP) to describe gases. Scientists have designated STP as being 1 atm and 0 C or 273 K. Many gas law problems use STP. The ideal gas law combines Boyle’s, Charles , and Gay-Lussac’s Law into one single equation. An ideal gas is an imaginary gas whose particles are infinitely small and do not interact with each other. The law works best at room temperature and atmospheric pressure The Ideal Gas Law is: PV=nRT Where P=pressure, V=volume in L, n=moles, R=Gas constant, and T=temperature in Kelvin. |
Practice
|
When you feel that you have covered an adequate amount of material in this section, then try the following problems:
Answer Keys: |
Watch Out for Those Tracker Jackers!
Extra Resources
Still confused? Check out these awesome sites:
- Brightstorm Gas Videos
- Khan Academy
- pHet Gas Law Simulation (Play with the pressure, temperature and volume to examine how each impact the motion of the gas molecules)