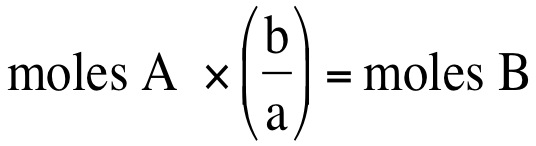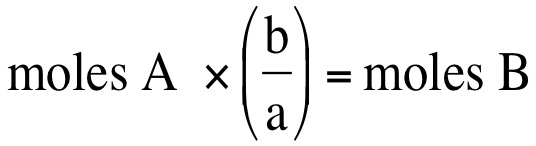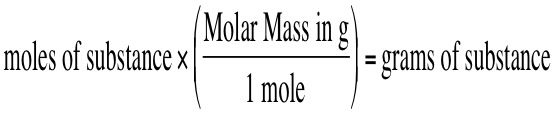District 5 - Stoichiometry and Solutions

The Capitol is so infatuated with stoichiometry and solutions that they granted them two Districts (as opposed to one that all other Districts are allotted). Additionally, the Capitol showers them with praise throughout the year by providing them with more food and supplies then all the other Districts. This District is just as fierce and destructive as their sister District (District 4). Many Hunger Games have come down to a battle royale between District 4 and 5. You will need to have a profound understanding of stoichiometry and solutions in order to take this District down because they possess the power to perform the ultimate kill move: the grams to moles to moles to grams conversion.
Your Training
Training Resources
Intermolecular Force
|
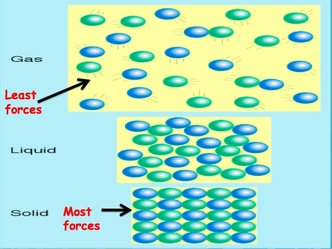
Atoms in a solid hold a very fixed position. They are said to be vibrating in place. There is a large amount of forces between the molecules.
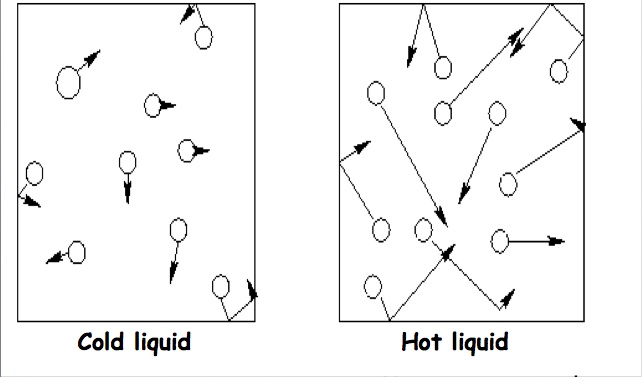
Atoms in a liquid are further spread out from one another. They flow freely within the solution and are moving in a random motion. There are some forces between the molecules, but not as many as solids have. Atoms in a gas are in constant random motion. There are very few forces between the molecules. Intermolecular forces are forces that attract molecules to one another. All intermolecular forces are the result of the attraction between opposite charges. The three main types of intermolecular forces are:
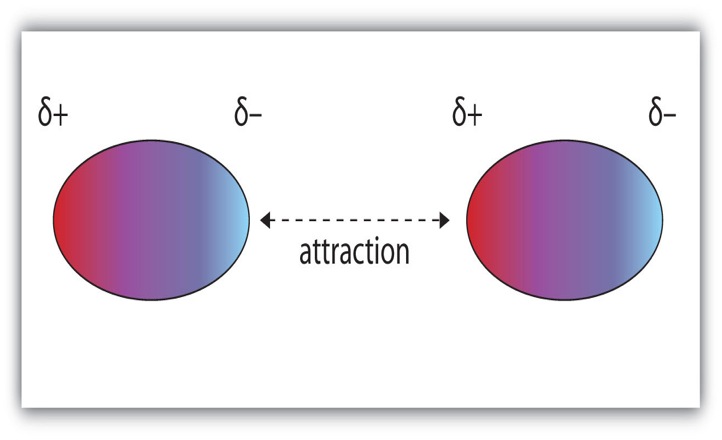
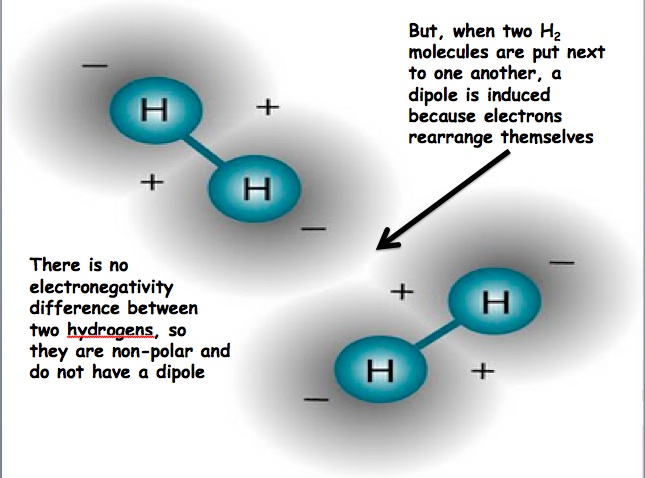
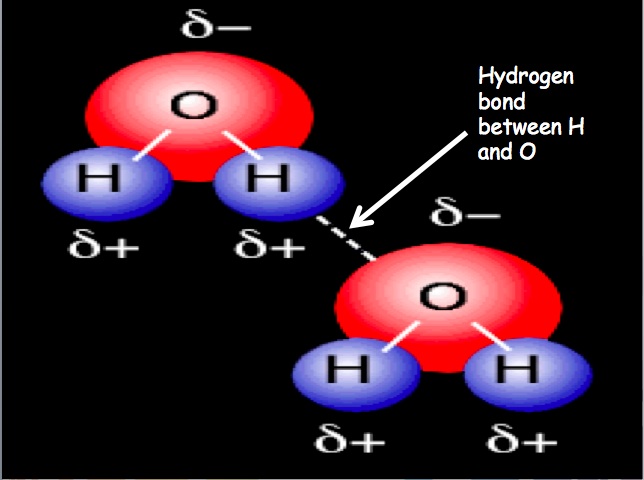
A dipole is a separation of charge. Recall, electronegativity is how well an element pulls electrons towards itself. Molecules can have a separation of charge because a very electronegative element is bound to an element with a lower electronegativity. This causes the electrons to be held closer to the element with the higher electronegativity. Dipole-Dipole Forces: Dipole-dipole forces refer to the attraction between molecules that have a permanent dipole. The positive end of one molecule will be attracted to the negative end of the other molecule. Molecules that have permanent dipoles are known as polar compounds. Dispersion Forces: Some molecules do not have dipoles within the molecule (e.g. O2), but they can have an induced dipole. Dispersion forces are the intermolecular forces resulting from the uneven distribution of electrons and the creation of temporary dipoles. This is the weakest intermolecular force. When two molecules are brought close to one another, electrons rearrange themselves so that a temporary dipole results, this is dispersion forces. Molecules that only exhibit temporary dipoles are known as non-polar compounds. Hydrogen Bonding: Hydrogen bonding refers to the attraction of one molecule that contains hydrogen to another molecule. When hydrogen is bound to N, O, or F, there is a large difference in electronegativity and this creates a partial positive charge and a partial negative charge (H has the partial positive charge because it has its electrons pulled away). The partial positive charge on hydrogen is attracted to N, O, or F of another molecule. This results in a hydrogen bond. |
|
Ways to Impact Solubility
|
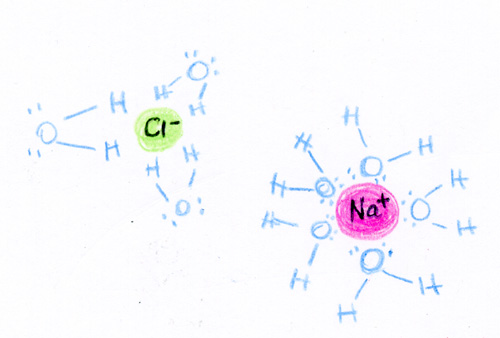
In order to dissolve a solute in a solvent, the solvent must use intermolecular forces in order to break the solute apart and pull it into solution.
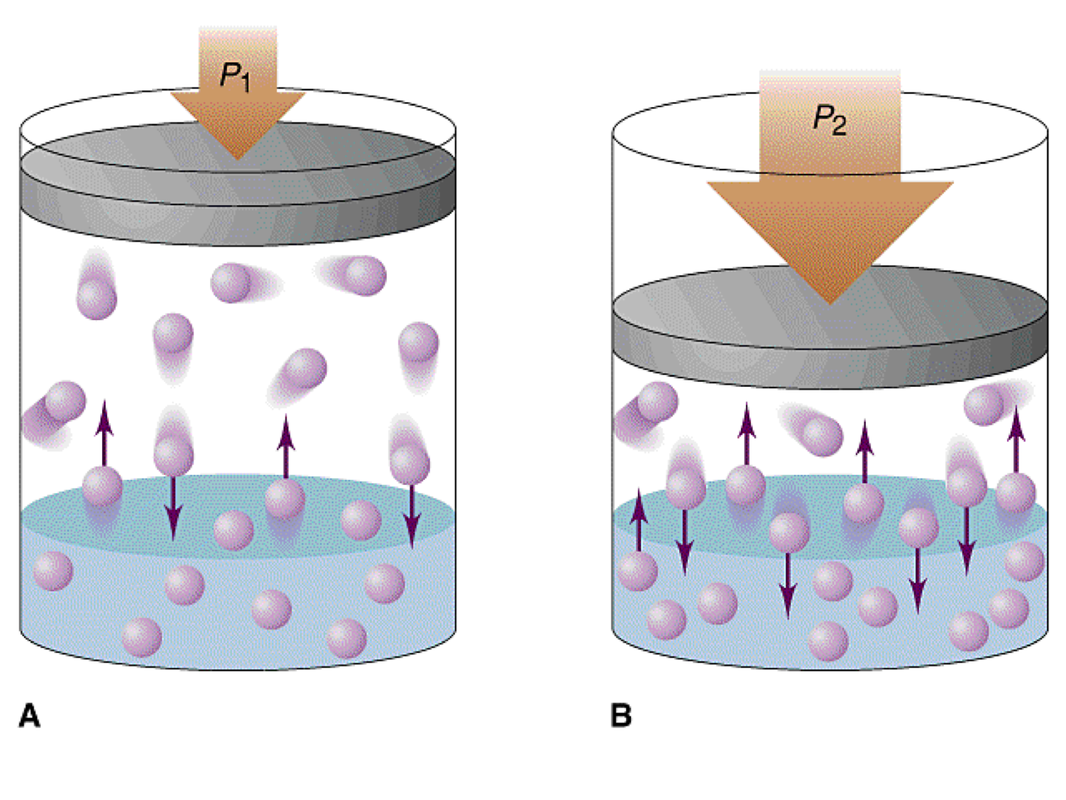
The most common example is NaCl dissolving in water. A water molecule has a dipole with a partial negative charge on the oxygen and a partial positive on the hydrogen. In addition to the hydrogen bonding that it can form with itself, it also can form dipole-dipole forces with NaCl. The negativly charged oxygen is attracted the positive Na ion and the positively charges hydrogen is attracted to the negative Cl. The water molecules form "water cages" around each Na and Cl ion in order to break the crystal apart and pull the solute into solution. The rate at which a solute dissolves in a solvent depends on numerous factors. We can alter the rate at which the solute dissolves by altering the temperature of the solution, the surface area of the solute, or the pressure of the solution. As you increase the temperature, the rate at which the solute dissolves increases. As you increase the temperature, molecules start moving faster. By moving faster, the molecules are better able to grab the solute, form the cages around them and pull them into the solution. Therefore, the faster molecules are moving, the faster something dissolves. As you increase the surface area, the rate at which the solute dissolves increases. As you increase the surface area of a substance, you are providing more sites for the solvent to interact with the solute. This allows for the solute to be pulled into solution at a much quicker rate when compared to those of a lower surface area. As you increase the pressure, the rate at which the solute dissolves increases. The solubility and pressure relationship is clearly seen when a gas dissolves in a liquid. When a gas is above a liquid and the pressure is increased, the gas molecules collide more often with the wall of the liquid and this causes more gas to be pushed into the liquid. |
|
Converting Moles to Moles
|
We can convert between moles of one substance and another substance. If we have a balanced chemical equation, then we can use the coefficients (numbers in front of each element/compound of a balanced chemical equation), then we can convert moles of one substance to moles of another substance. If we have the general equation:
aA + bB --> cC + dD where lowercase letters represent coefficients and upercase letters represent elements/compounds, then we can convert moles of one substance to moles of another substance: Moles A represents the amount of moles that you are starting with and b/a represents the ratio of the coefficients from the balanced chemical equation or the mole ratio.
There are two important things to remember when using this:
|
Grams to Moles to Moles to Grams: The Ultimate Kill Move
|
We now are able to combine all of the conversions that we have talked about in both District 4 and 5 in order to perform the ultimate kill move: the grams to moles to moles to gram. This move has struck fear into the hearts of many tributes, but if you can remember the basics you will begin to recognize that you too can complete this move.
Recall, in order to covert grams of some substance to moles of some substance we need to utilize the molar mass. If you have grams of some substance and want to convert to moles, then you use the equation: Once you have moles of that substance, then you can convert to moles of another substance by using the mole ratio. For the general equation:
aA + bB --> cC + dD where lowercase letters represent coefficients and uppercase letters represent elements/compounds. Therefore, if we have moles of substance A and want to convert to moles of substance B, then we use the equation: Once you have moles of this new substance, then you convert them to grams of this new substance. You just need to do the reverse of what you did for the first equation in this series. Once you have moles of this new substance, then you can use the following equation to convert to grams of this new substance:
If you are able to execute these 3 equations, then you will be able to complete the ultimate kills move: the grams to moles to moles to grams. You will be a heavy favorite to win the Games if you can master this move.
Quick Practice! |
Get That Arrow Ready!
Extra Resources
Still confused? Check out these awesome sites:
- Brightstorm Stoichiometry Videos
- Brainpop Videos (ask Mr. Lance for login information)
- Khan Academy Videos