District 3 - Chemical Bonding

Welcome to your training for how to take down District 3 when you are in the arena. The elements of District 1 and 2 have nothing on these guys. This is the place where all the elements are combined, broken apart, and recombined in an infinite number of arrangements. From this District, we get all the compounds that exist within our known universe. They are advised by past champion (and man who is on constant suicide watch) Gilbert Lewis. You will need to be beware of District 2's ability to mix elements together to make a nasty compound that will do terrible (and likely irreversible) damage to you.
Past District 3 Champion
Your Training
Training Resources
Ionic Bonding
Please take out your District 3 guided notes and watch the lecture below.
|
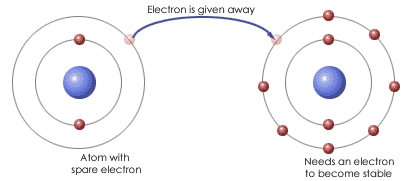
Electrons are often transferred between elements so that both of them are able to complete their octet. An element that wants to get rid of electrons gives away its electrons to an element that wants to gain electrons to complete its octet. This transfer of electrons causes an ionic bond to form.
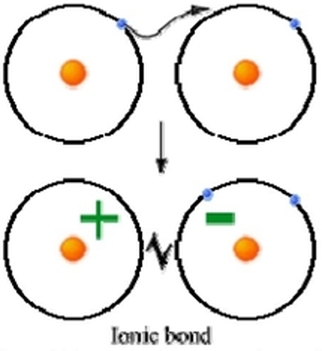
Metals typically donate or give away their electrons in order to achieve a full outer valence. Non-metals typically accept electrons in order to achieve a full outer valence. This exchange of electrons leads to an ionic bond. Ionic Bond – A bond that forms between an anion and cation as a result of the elements exchanging electrons. A monoatomic ionic bond is a bond that forms between 1 positive element and 1 negative element. Typically a bond between a metal and a non-metal. These elements bond to form a compound. A compound is any substance made of atoms of two or more different elements joined by a chemical bond. This compound does not have a charge because ions combine in order to create a final charge of 0. To determine the chemical formula for an ionic compound you can follow the following steps:
To write out polyatomic ionic compounds follow the following steps:
|
Crystal Lattices
Please take out your District 3 guided notes and watch the lecture below.
|
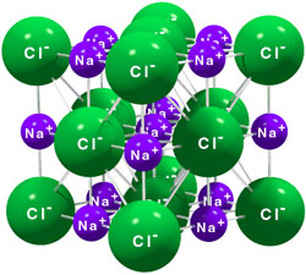
Ionic compounds have a positive and negative charge and this charge causes salt crystals or crystal lattices to form. Salt crystals that we see are repeating structures of a very simple unit. Arranged so that there is a repeating structure of positive and negative charges
Crystal lattices have a structure of repeating positive and negative charges Positive charged ions in an ionic compound are attracted to other negative charged ions. This attraction between positive and negative charges holds crystal lattices together. This attraction between positive and negative charged ions is known as electrostatic attraction. |
Covalent Compounds and Lewis Structures
Please take out your District 3 guided notes and watch the lecture below.
|
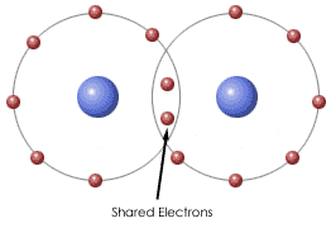
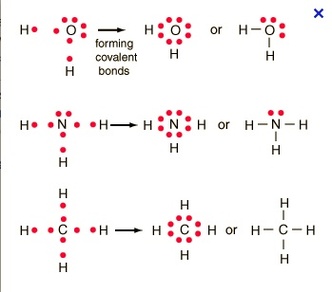
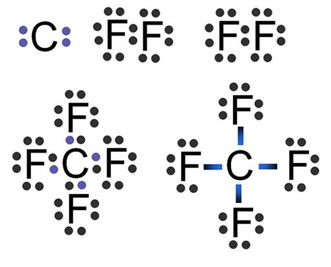
Covalent bonds are a bond between two or more non-metals. In a covalent bond, the electrons are shared between the elements so that each can achieve a full outer valence shell.
Identifying ionic and covalent compounds can be done by examining the elements that make up the compound. If it is made of a metal and a non-metal, then it is an ionic compound. If it is made of two or more non-metals, then it is a covalent compound. Most molecules in biology are covalent compounds. Things such as proteins, carbohydrates, and DNA all are held together by the sharing of electrons. |
|
Lewis Dot structures for an element are like Bohr diagrams, but they only show the valence electrons. The group number (Group 1A, 2A, etc.) tells us the number of valence electrons for elements.
The steps to draw a Lewis structure for an element are as follows:
When drawing Lewis structures there are a few guiding rules to help you draw the correct molecule:
|
Practice Questions
|
Complete the following practice problems in order to train to take down the District 3 tributes:
Answer Key |
Gilbert Lewis' Plan
|
As you begin to eat the small amount of food you have found, you smell a hint of almond and drop the food. You realize that this is the doing of District 3 and their master chemist Gilbert Lewis. In order to ensure that you can use this trick to kill District 3, complete the following problems:
|
Extra Resources
Still confused? Check out these awesome sites:
- Brightstorm Bonding Videos
- Brainpop Videos (ask Mr. Lance for login information)
- Khan Academy Videos
- pHet Lewis Structure Simulator (Check out the other simulators that may help!)